Many students taking Orgo 1 have commented there are a few types of reactions the professors save to the end of the semester and cover quickly and “gloss” over or sometimes skip all together in the interest of time. However, in Orgo 2, you will be responsible for all of the reactions necessary for multi-step synthesis (starting product known to get to unknown final product) and retrosynthesis (product known to get to unknown starting material) reactions. We at StudyOrgo don’t want you to get stuck on trying to cram for exams by studying reactions that were poorly covered in your class. In this article, we focus on epoxide formation and epoxide ring-opening reactions because of their usefulness in synthesis and industrial application.
Epoxide Formation
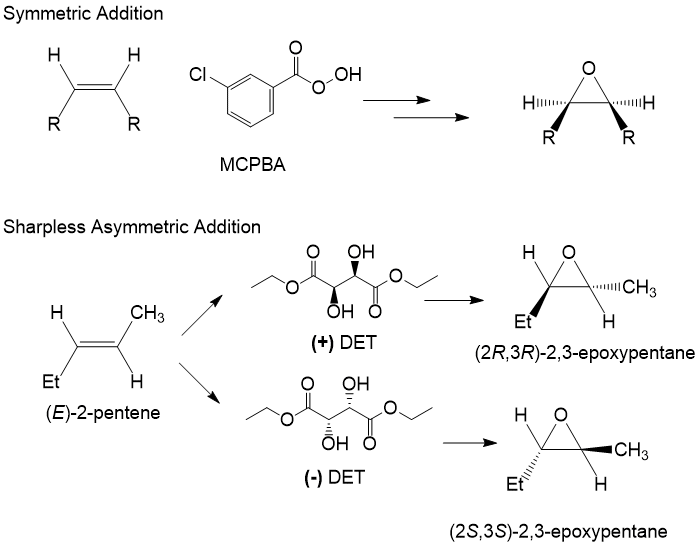
In order to form an epoxide, a electron-rich reagent is required, such as an alkene. Formation of the epoxide occurs in the presence of a peroxide reactant, such as MCPBA or DET. The choice of reagent depends on the stereo-specificity desired from the reaction. MCPBA is used to add the epoxide symmetricaly across the double bond, so that substituents that are cis- or trans- remain in this configuration. The Sharpless asymmetric addition allows the researcher to choose a stereoisomer of DET, referred to as (+) and (-). This reagent allows for the attack of the peroxide intermediate on only one face of the alkene, allowing for the production of nearly pure enantiomeric excess product, generally >98%!
Ring-opening Reactions
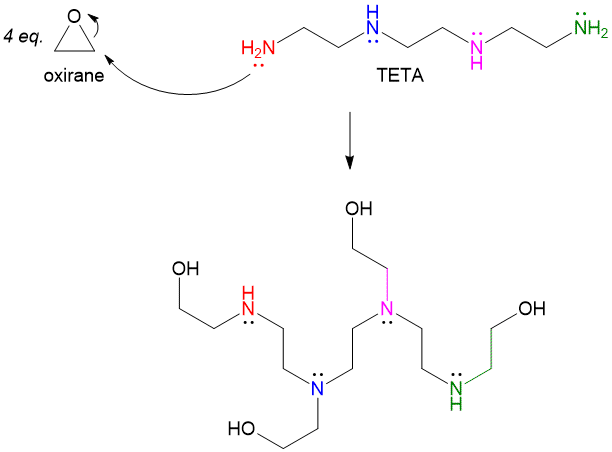
Many of you have probably hear of epoxy-glue, which is a very strong binding agent. The process usually involves mixing two reagents and you must quickly apply the mixture to the broken items before the expoxy hardens. One tube will contain the resin, or epoxide, while the other contains the “hardening” agent, which is the nucleophile that will attack the epoxide. In a ring-opening reaction, a molecule such as TETA, which contains 4 amino groups, will attack 4 equivalents of oxirane to produce a complex polymer, which is the basis for a strong glue. Changing the size and complexity of the epoxide can allow for flexibility of strength, thermostability and rigidity!
We here at StudyOrgo have devoted countless hours to preparing complex reaction mechanisms in simple and easy-to-understand manner to help you maximize your studying. Sign up with StudyOrgo for detailed explanations of epoxide reaction mechanisms and other essential Orgo 2 reactions today!