This is the final part of a four part series in the Energy Diagram Module.
Click on the following link to see earlier parts:
Part 1
Part 2
Part 3
For each of the following questions- choose the best answer among the four answer choices.
Scroll to the bottom to see the answer key.
Question1
The net amount of heat released from the formation of products in a reaction is represented by _______________.
A) Temperature
B) Activation Energy
C) Heat of Reaction
D) Entropy
Question 2
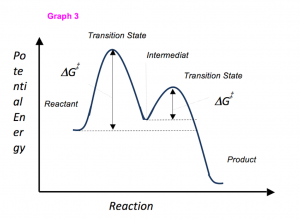
A transition state represents _______________.
A) The highest potential energy molecule of the mechanism
B) The lowest potential energy molecule of the mechanism
C) The a molecule that does not occur in the reaction
D) A new reactant introduced to the reaction.
Question 3
A high energy, sometimes isolatable, chemical species during the course of reactants to products is referred to as a _______________.
A) Product
B) Midpoint
C) Transition State
D) Intermediate
Question 4
Refer to the Gibbs Free Energy Equation
ΔG0 = ΔH0 – TΔS
Under which circumstance will DG0 NEVER be spontaneous. (Hint: spontaneous reactions have a negative ΔG0)
A) ΔH0 < 0 and ΔS < 0
ΔH0 > 0 and ΔS < 0
C) ΔH0 > 0 and ΔS > 0
D) ΔH0 < 0 and ΔS > 0
ANSWER KEY:
1. C
2. A
3. C
4. B